 Which unit cell has the minimum or lowest packing efficiency? Packing Fraction Formula = Volume Occupied by all constituent particles Total Volume of Unit CellUnit Cell Packing Efficiency – VEDANTUwww. 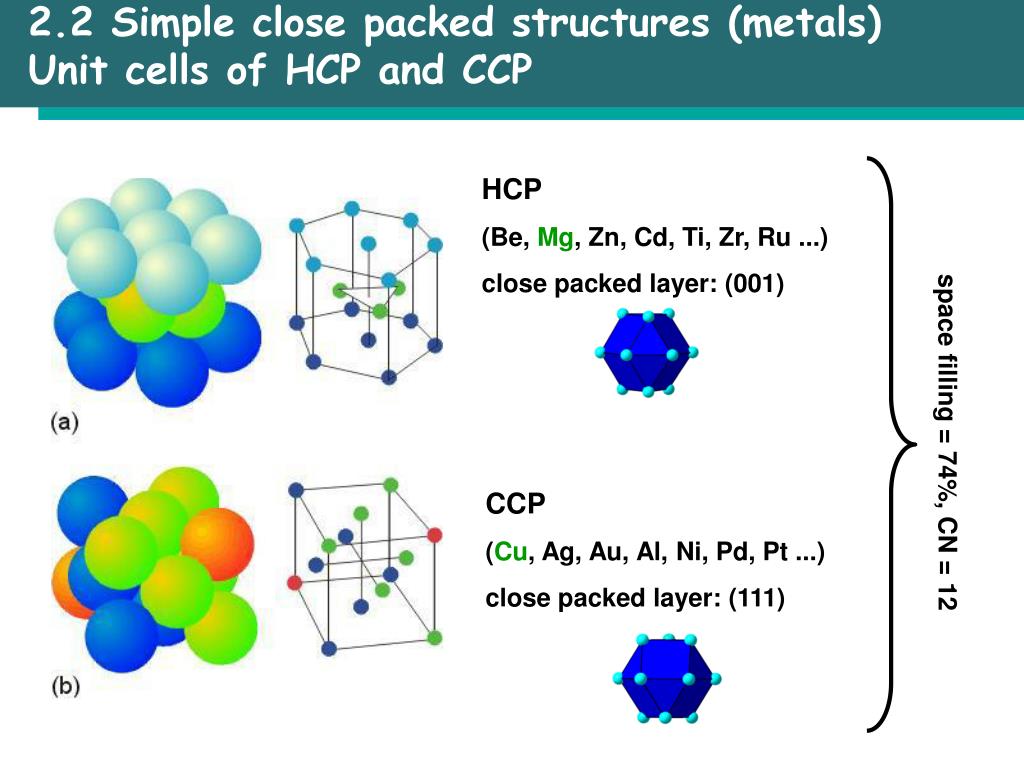
Packing Efficiency Formula = No of Atoms x Volume Occupied by 1 atom Total Volume of Unit Cell X 100 We can say that Packing Fraction, when multiplied by 100, is seen as a percentage, it becomes the Packing Efficiency of that particular cell. The packing efficiency of simple cubic unit cell is 52. The packing efficiency of Bcc unit cell is 68%. The packing efficiency of Fcc unit cell is 74%. The packing efficiency is the percentage of total space occupied by the particles. What is the packing efficiency of simple cubic unit cell? What is the packing efficiency of BCC and CCP? The volume occupied by 4 atoms is 4Ã-34‹Ïr3=316‹Ïr3 The packing efficiency =total volume of unit cellVolume occupied by atoms in a unit cell ‹Ã-100 The packing efficiency =88‹r3316‹Ïr3‹Ã-100=74. Let a unit cell of bcc structure with side a.What is the packing efficiency of a CCP unit cell?Ī CCP unit cell has 4 atoms per unit cell. Therefore, total number of atoms present in bcc unit cell is equal to 2. 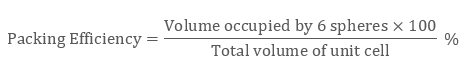
In body centered cubic unit cell, one atom is present in body center apart from 4 atoms at its corners. Thus,packing efficiency of hcp or ccp structure=74% Packing efficiency of body centered cubic (bcc) structure: Now, packing efficiency (in %) `=text(volume of 4 spheres in unit cell)/text(total volume of unit cell)xx100` Since one unit cell of ccp or hcp contains 4 atoms, i.e. Substituting the value of a from equation (i) we get `= text(Volume of 4 spheres in unit cell)/text(Total volume of unit cell)xx100`ĪB is perpendicular, DC is base and AC is diagonal Therefore, packing efficiency of hcp or ccp structure Since there are 4 atoms in the unit cell of hcp or ccp structure 
Thus, packing efficiency (in %) `= text(Volume of spheres in unit cell)/text(Total volume of unit cell)xx100` The packing efficiency can be calculated by the percent of space occupied by spheres present in a unit cell. Solid State Solutions Electrochemistry Chemical KineticsĬalculation of pacing efficiency in hcp and ccp structure: And the packing efficiency of body centered cubic lattice (bcc) is 68%. 
The packing efficiency of simple cubic lattice is 52.4%. The hcp and ccp structure are equally efficient in terms of packing. 74% of the space in hcp and ccp is filled. The packing efficiency of both types of close packed structure is 74%, i.e. Solid State Packing Efficiency of Close Packed Structure - 1īoth ccp and hcp are highly efficient lattice in terms of packing. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
